
-
Recent Posts
Archives
Categories
Meta

A new edited volume is coming out that is focused on the ecological interactions of caterpillars, their host plants, their mutualists, and their enemies. Robert Marquis and Suzanne Koptur (the editors) have collected chapters on a diverse array of topics involving caterpillars and their interactions with other species authored by dozens of top-notch scientific researchers. Throughout the book there is an emphasis on how these interactions are affected by anthropogenic changes to abiotic and biotic environments. I want to highlight one chapter in particular authored by Scott Shaw (U. Wyoming) and myslef focused on the the natural history and ecology of caterpillar parasitoids! I will try to announce when the volume is officially published (but given my recent record with updating this site, I may be a bit late with that!).

Today a paper headed by Silvia Gisondi (a graduate student in Italian tachinid expert Pierfilippo Cerreti’s lab) is now available. It describes the first male of the unusual European tachinid, Pelamera atra, and, based on molecular phylogenetic data (see tree below), establishes a new monotypic tribe for the genus. Congratulations to Silvia! The full paper can be accessed at this link for the next 50 days https://authors.elsevier.com/c/1c69JA7vOnHH1 (of course we would also be happy to send a pdf version upon request).
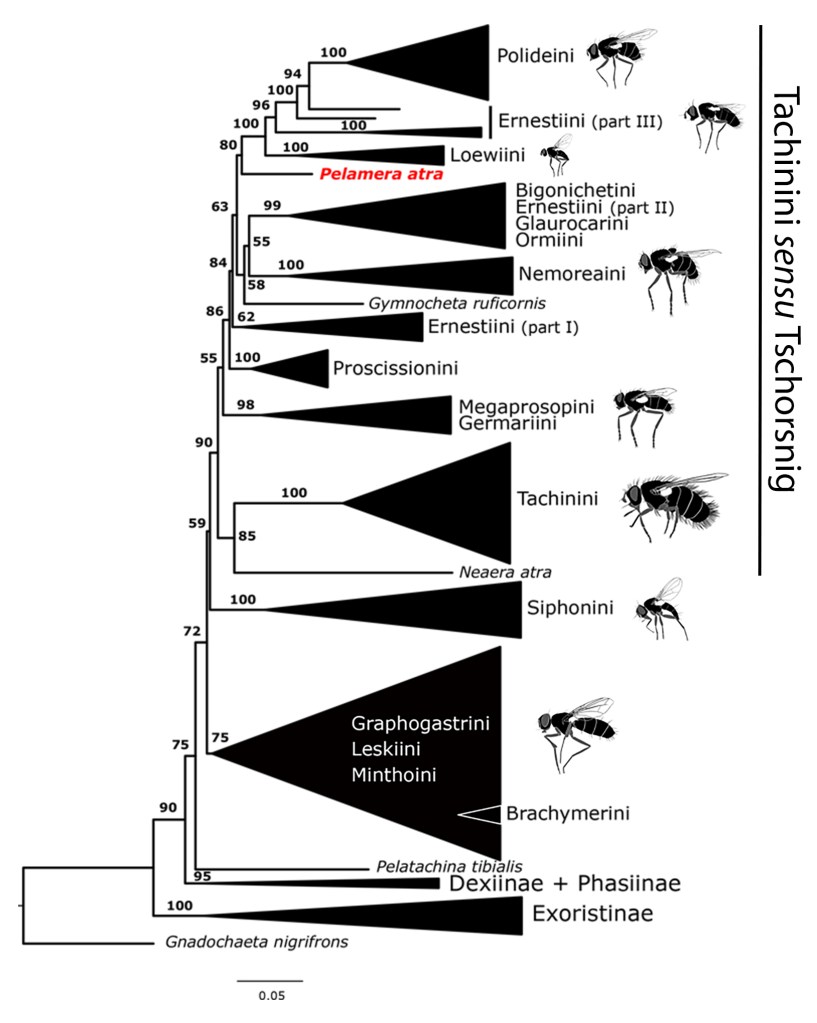
A phylogenetic tree showing the position of Pelamera atra in the Tachinini sensu lato
I have been quite lax in providing updates and posts to the Stireman lab website. Hopefully, I will be able to devote time to it in the future. A number of publications have come out of the Stireman lab since I last posted two years ago. Most notable among them is A Molecular Phylogeny of World Tachinidae which was published online in early 2019 (Stiremanetal2019YMPEV6358.pdf), which was the culmination of a number of years’ work by me (Stireman), Jim O’Hara, Pierfilippo Cerretti, Kevin Moulton, and Jeremy Blaschke. I hope that others find this paper interesting and useful. It is by no means the “end of the story” regarding tachinid phylogeny, but it is a significant advancement that we hope will stimulate more research into understanding this evolution and diversification of this fascinating group. Abstract below.
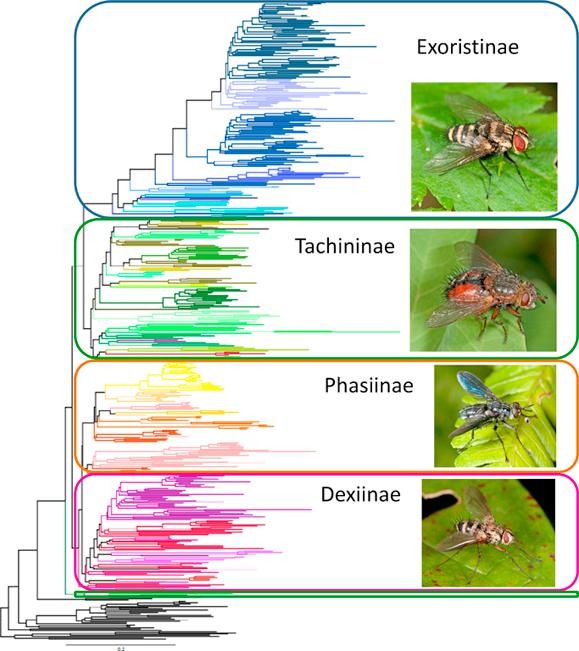
We reconstructed phylogenetic relationships within the diverse parasitoid fly family Tachinidae using four nuclear loci (7800 bp) and including an exceptionally large sample of more than 500 taxa from around the world. The position of the earthworm-parasitizing Polleniinae (Calliphoridae s.l.) as sister to Tachinidae is strongly supported. Our analyses recovered each of the four tachinid subfamilies and most recognized tribes, with some important exceptions in the Dexiinae and Tachininae. Most notably, the tachinine tribes Macquartiini and Myiophasiini form a clade sister to all other Tachinidae, and a clade of Palpostomatini is reconstructed as sister to Dexiinae + Phasiinae. Although most nodes are well-supported, relationships within several lineages that appear to have undergone rapid episodes of diversification (basal Dexiinae and Tachininae, Blondeliini) were poorly resolved. Reconstructions of host use evolution are equivocal, but generally support the hypothesis that the ancestral host of tachinids was a beetle and that subsequent host shifts to caterpillars may coincide with accelerated diversification. Evolutionary reconstructions of reproductive strategy using alternative methods were incongruent, however it is most likely that ancestral tachinids possessed unincubated, thick shelled eggs from which incubated eggs evolved repeatedly, potentially expanding available host niches. These results provide a broad foundation for understanding the phylogeny and evolution of this important family of parasitoid insects. We hope it will serve as a framework to be used in concert with morphology and other sources of evidence to revise the higher taxonomic classification of Tachinidae and further explore their evolutionary history and diversification.
Finally, the last paper stemming from Jeremy Heath’s PhD research has been published in The American Naturalist! This is a super-cool paper illustrating the importance of natural enemies (parasitoids in this case) in driving the adaptive diversification in their gall midge hosts. We suggest that such top-down driven diversification (rather than resource-driven) may be much more widespread than is currently appreciated. I could explain further, but I think the abstract below sums it up nicely. You can access the paper here.

Aprostocetus tesserus laying an egg in an Asteromyia carbonifera gall. (J.J. Heath)
Abstract
Most studies of adaptive radiation in animals focus on resource competition as the primary driver of trait divergence. The roles of other ecological interactions in shaping divergent phenotypes during such radiations have received less attention. We evaluate natural enemies as primary agents of diversifying selection on the phenotypes of an actively diverging lineage of gall midges on tall goldenrod. In this system, the gall of the midge consists of a biotrophic fungal symbiont that develops on host-plant leaves and forms distinctly variable protective carapaces over midge larvae. Through field studies, we show that fungal gall morphology, which is induced by midges (i.e., it is an extended phenotype), is under directional and diversifying selection by parasitoid enemies. Overall, natural enemies disruptively select for either small or large galls, mainly along the axis of gall thickness. These results imply that predators are driving the evolution of phenotypic diversity in symbiotic defense traits in this system and that divergence in defensive morphology may provide ecological opportunities that help to fuel the adaptive radiation of this genus of midges on goldenrods. This enemy-driven phenotypic divergence in a diversifying lineage illustrates the potential importance of consumer-resource and symbiotic species interactions in adaptive radiation.
 Stireman (me) was recently invited to write a mini-review of the community ecology of non-hymenopteran parasitoids for the journal Current Opinion in Insect Science. This was a difficult task as the review was supposed to focus on recent (last 5 years) literature, and was severely constrained in length and number of references. Still, I hope that it provides a decent overview of some of the recent research on the topic (focusing on dipteran parasitoids) and helps to spur further research.
Stireman (me) was recently invited to write a mini-review of the community ecology of non-hymenopteran parasitoids for the journal Current Opinion in Insect Science. This was a difficult task as the review was supposed to focus on recent (last 5 years) literature, and was severely constrained in length and number of references. Still, I hope that it provides a decent overview of some of the recent research on the topic (focusing on dipteran parasitoids) and helps to spur further research.
The full text can be accessed here at the publisher’s web site (at least for a while). Enjoy!
I am currently se eking a Ph.D. student to join my laboratory studying the evolution and ecology of parasitoid flies. While the specific focus of the dissertation research is negotiable, the research assistantship will require contributing to a collaborative, NSF/Brazil(FAPESP) funded Dimensions of Biodiversity project focused on “Chemically mediated multi-trophic interaction diversity across tropical gradients.” My laboratory’s role in this international collaborative project is primarily focused on tachinid parasitoids. This includes identifying and documenting species, studying how they influence and are influenced by hosts and their host-plants, analyzing population- and phylo-genetic/genomic patterns and processes, and revisionary taxonomy and species description. Students will also have the opportunity to contribute to other aspects of this large and multi-disciplinary project . The successful applicant will develop a thesis research project on tachinid ecology, evolution and/or systematics employing ecological, phylogenomic, taxonomic, and comparative methods. The student will also have the opportunity to visit and participate in field research in Brazil as well as other Latin American Countries. Latin American students are particularly encouraged to apply. Click here for more information.
eking a Ph.D. student to join my laboratory studying the evolution and ecology of parasitoid flies. While the specific focus of the dissertation research is negotiable, the research assistantship will require contributing to a collaborative, NSF/Brazil(FAPESP) funded Dimensions of Biodiversity project focused on “Chemically mediated multi-trophic interaction diversity across tropical gradients.” My laboratory’s role in this international collaborative project is primarily focused on tachinid parasitoids. This includes identifying and documenting species, studying how they influence and are influenced by hosts and their host-plants, analyzing population- and phylo-genetic/genomic patterns and processes, and revisionary taxonomy and species description. Students will also have the opportunity to contribute to other aspects of this large and multi-disciplinary project . The successful applicant will develop a thesis research project on tachinid ecology, evolution and/or systematics employing ecological, phylogenomic, taxonomic, and comparative methods. The student will also have the opportunity to visit and participate in field research in Brazil as well as other Latin American Countries. Latin American students are particularly encouraged to apply. Click here for more information.
Congratulations are in order! WSU master’s student Karen Pedersen has successfully defended her thesis research on: LIMITATIONS OF HOST PLANT USE IN TWO ANDEAN ALTINOTE (NYMPHALIDAE, HELICONIINAE, ACRAEINI), BUTTERFLIES, FROM A TRITROPHIC PERSPECTIVE. In this research project, Karen tried to assess the determinants of host plant use in two co-occurring species of Altinote butterflies in Ecuador. Her research took place at Yanayacu Biological Station, and is an extension of our work there on plant-caterpillar-parasitoid interactions. Karen produced several short videos focused on her study system system that you can check out:
Although MS student Diego Inclan successfully defended his thesis more than two years ago, the focus of his research was published this year:
Inclan, D.J. & Stireman, J. O. III. 2013. Revision of the genus Erythromelana Townsend, 1918 (Diptera: Tachinidae) with description of 11 new species and analysis of their phylogeny and diversification. Zootaxa. 3621:1-82.(Abstract here)
In this lengthy manuscript Diego describes 11 new species (as you can tell from the title) of a little known, small bodied and somewhat gracile genus of tachinids in the tribe Blondeliini that occur in the Neotropical region (Northern Argentina
to Southern Mexico). Most of the species are found in cloud forest habitats in the Andes. Diego also examines their phylogenetic relationships using morphological and molecular approaches and examines potential modes of diversification for the genus. With only three previously described species, Diego has more than quadrupled the number of known species in the genus.
Why focus on this group of small, poorly known cloud forest tachinids?
First, exploring, documenting and describing all of Earth’s organisms is a worthy goal in itself. Each has many fascinating biological “stories” or “lessons” to tell with its unique evolutionary history and ecological niche. Describing these species is a first step towards elucidating these lessons, which may be far reaching (e.g., I always like to point out the vast scientific knowledge we have gleaned from studying the small and nondescript species Drosophila melanogaster).
However, there is another reason that we focused on this genus. It just so happens that Erythromelana species are parasitoids of small geometrids in the genus Eois. We know this because we reared several species (first known rearings for the genus) from Eois in the cloud forests of Ecuador (see post Ecuador Expedition). Eois is a very diverse genus of geometrids that feed on plants in the hyper-diverse genus Piper (Piperaceae), and this tritrophic system of plants, herbivores, and parasitoids has been the subject of a number of studies by collaborators of our “Caterpillars and Parasitoids of the Eastern Andes” biological survey project (e.g., see Wilsonetal2011). One goal of Diego’s paper was to see if Erythromelana exhibited evidence of co-evolution and host-associated differentiation with their Eois hosts. Although the degree of specificity of Erythromelana species is difficult to evaluate due to the many undescribed and cryptic species of Eois that we know of, our rearing records suggest considerable overlap in host use among these tachinids. Thus, it would appear that perhaps geographic isolation may play a greater role in Erythromelana diversification than host associations.
Why ‘ghosts of the cloud forest’? This somewhat dramatic title refers to the rarity of Erythromelana species. Total parasitism of Eois (from over 5000 rearings) is less than 0.5% and we only recovered a single specimen from a pan trapping effort at Yanayacu that collected over 2000 other tachinids. Several of the described species are known from just a handful of specimens. Still, these ‘ghosts’ can be observed, if one knows where to look. On a few occasions they have been seen, flitting about sunlit leaves over small streams in deep cloud forests of Costa Rica and Ecuador.
Just a few recent photos from my yard (click for larger images)